Screening for HPV 16, 18 subtypes of high-risk. Early screening for cancer. This product is designed for the in vitro characterization of human papillomavirus L1 envelope proteins, including HPV-16, -18, in female cervical secretion samples. Human papillomavirus (HPV) infection is the main causative factor of cervical cancer in women. Cervical cancer is a common malignant tumor in women, with a peak incidence age of 30-50 years. About 110 types of HPV have been identified, which are classified into high-risk and low-risk types according to their incidence and carcinogenicity to malignant tumors. Among them, there are 13 high-risk types (including HPV 16, 18 ,31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 68). The results of this kit can be used as an aid in the clinical diagnosis of HPV-16, -18 infections.
Infection with human papillomavirus (HPV) is a leading cause of cancer among women worldwide with approximately 500,000 new cervical cancer cases and 250,000 deaths each year. Cervical cancer is caused by persistent infection with a group of carcinogenic HPV genotypes (HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, and probably HPV68)
The importance of cervical cancer is accentuated by the relatively young average age at incidence and death.
Cervical cancer screening strategies have evolved from cytology-based to HPV-based. Following the identification of HPV as the cause of cervical cancer and the development of sensitive HPV tests, HPV-based screening permits the extension of screening intervals and increased impact per number of lifetime screens.
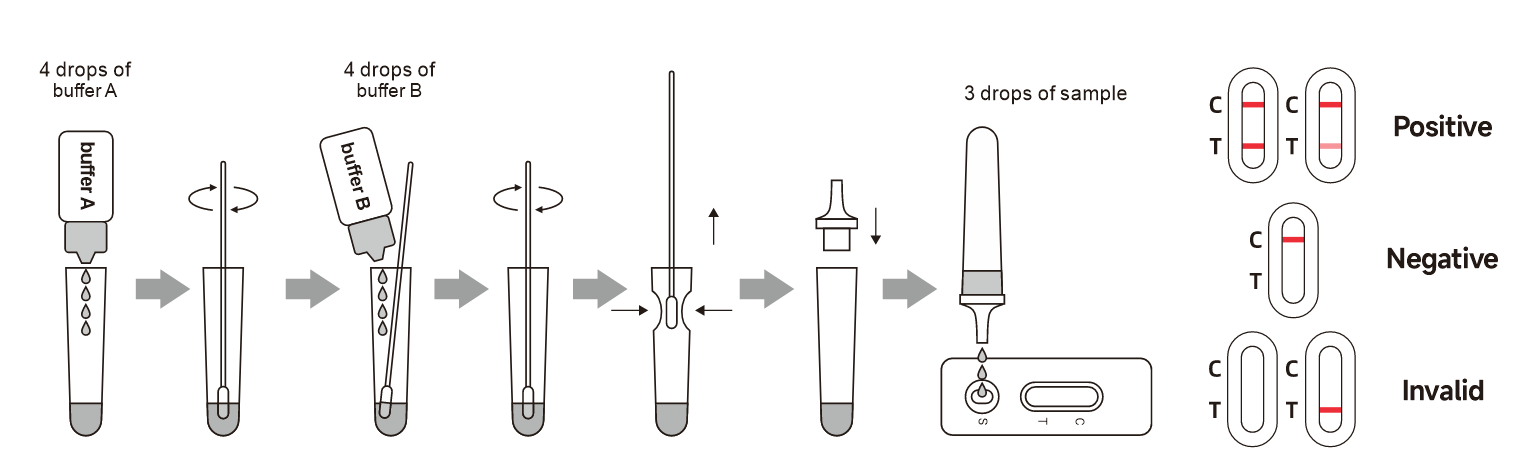
The Human Papilloma Virus (HPV) Antigen Rapid Test utilizes of specific monoclonal antibodies to qualitatively detect HPV L1 Capsid protein in female cervical swab specimen, the test can be performed without cumbersome laboratory equipment, and the results are available at 15 minutes.
The Human Papilloma Virus (HPV) Antigen Rapid Test is a lateral flow chromatographic immunoassay for the qualitative detection of HPV L1 Capsid protein in female cervical swab specimens. It is intended to be used as an aid in the diagnosis of cervical pre-cancer including types 16, 18. The test only provides preliminary screening results but not critical diagnosis criteria. Any reactive specimen with the Human Papilloma Virus (HPV) Antigen Rapid Test must be analyzed and confirmed with alternative testing method(s) and clinical findings. The test is intended for healthcare professional use.
| Product name | Human Papilloma Virus (HPV) Antigen Rapid Test Specification |
| Specification | Vaginal Secreta |
| Format | Cassette |
| Pack/Box | 25T |
| Shelf Life | 24 months |
| Storage temperature | 2-30℃ |
| Certificate |  |

To monitor your health status anytime, anywhere and achieve self-inspection and self-testing.