The COVID-19 Antigen Rapid Test Cassette is a lateral flow immunoassay intended for the qualitative detection SARS-CoV-2 nucleocapsid antigens in nasopharyngeal swab and nasal swab from individuals who are suspected of COVID-19 by their healthcare provider.
| COVID-19 (SARS-CoV-2) Antigen Test Kit | |
| Certificate system | CE certification |
| Sensitivity | 94.31% |
| Specificity | 99.21% |
| Accuracy | 96.98% |
The novel coronavirus belongs to the β genus. COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection: asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.
This product is used for the qualitative detection of novel coronavirus antigen infection in human nasal swab/nasopharyngeal swab samples
Using the principle of double antibody sandwich method
Simple: simple operation, easy to interpret
Fast: the detection is fast, the result can be interpreted in 15 minutes
Quick screening for early infection
Accuracy: high sensitivity and specificity
Stable: easy to store and transport
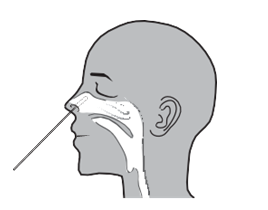
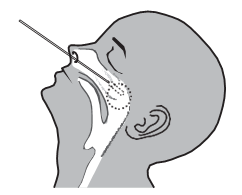
Operation A (nasal swab) Operation B (nasopharyngeal swab)
 |  |  |
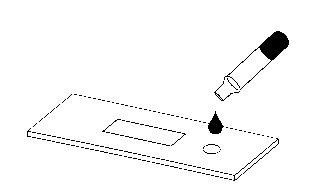 |  |  |
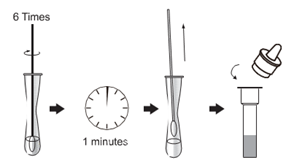
Add 3 drops of sample to be tested (about 120μL)
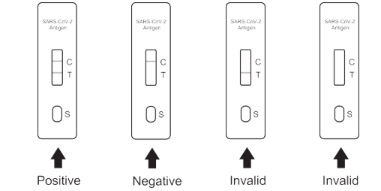
Positive (+): Two purple-red bands appear. One is located in the detection area (T), and the other is located in the quality control area (C).
Negative (-): Only a purple-red band appears in the quality control area (C). There is no purple-red band in the detection area (T).
Invalid: There is no purple-red band in the quality control area (C).

To monitor your health status anytime, anywhere and achieve self-inspection and self-testing.